A practical overview of molecular replacement: Clostridioides difficile PilA1, a difficult case study
Crawshaw AD, Basle A, Salgado PS
Acta Cryst D76, 2020
Abstract
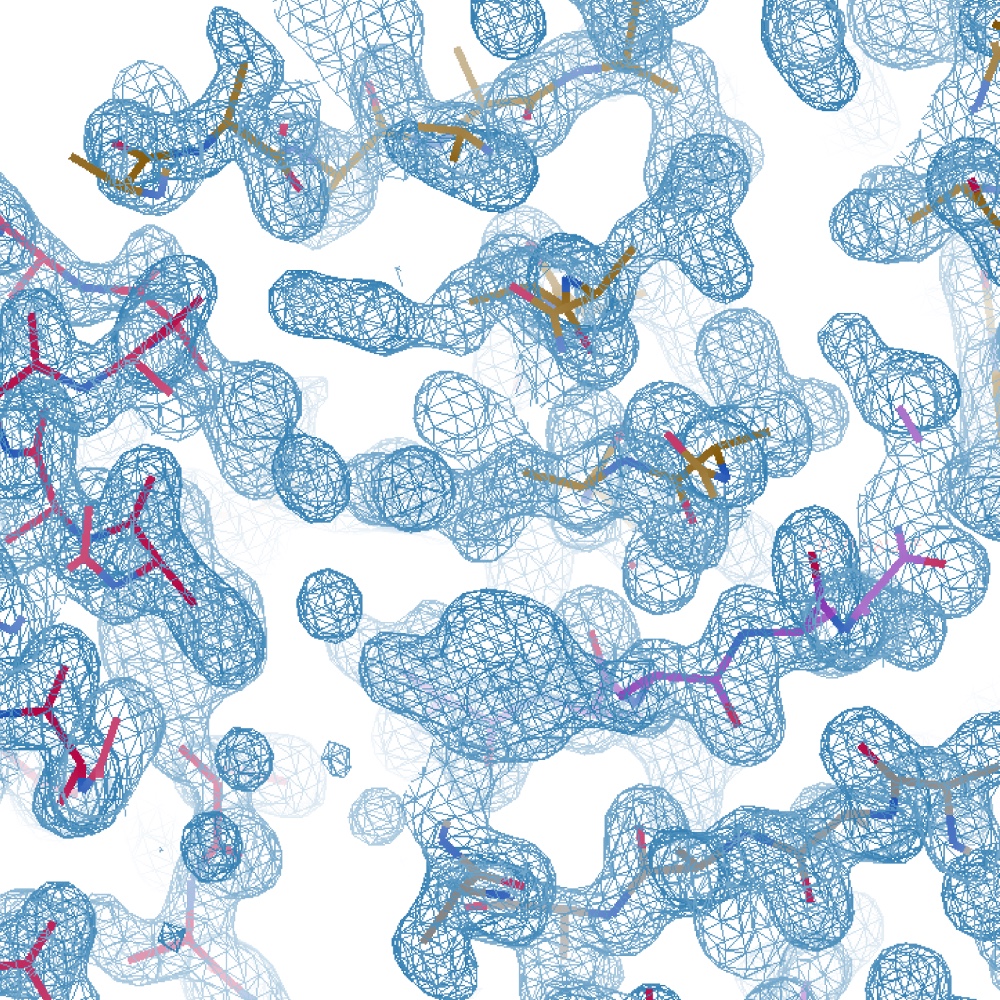
Many biologists are now routinely seeking to determine the three-dimensional structures of their proteins of choice, illustrating the importance of this knowledge, but also of the simplification and streamlining of structure-determination processes. Despite the fact that most software packages offer simple pipelines, for the non-expert navigating the outputs and understanding the key aspects can be daunting. Here, the structure determination of the type IV pili (TFP) protein PilA1 from *Clostridioides difficile *is used to illustrate the different steps involved, the key decision criteria and important considerations when using the most common pipelines and software. Molecular-replacement pipelines within CCP4i2 are presented to illustrate the more commonly used processes. Previous knowledge of the biology and structure of TFP pilins, particularly the presence of a long, N-terminal α-helix required for pilus formation, allowed informed decisions to be made during the structure-determination strategy. The PilA1 structure was finally successfully determined using ARCIMBOLDO and the ab initio MR strategy used is described.